 #color(blue)(t = ln((_0)/(_0))/(k(_0 - _0)))#Įven though we are doing half-life, we don't know their half-lives, so we can't assume that # = _0/2# or that # = _0/2#. In the interest of time (and partial fractions is not the focus here): Deriving half-life equation of a second-order reaction starting from the integrated rate law. With this, we need to use partial fractions to integrate the right side. Using separation of variables again, we have: Determine the half-life of the reaction when the concentration of the reactant. When the concentrations of both compounds decrease, they decrease by an unknown amount, #x#, so we have: The half-life of a second-order reaction is 124 s when the concentration of the reactant at the start is 0.0122 M. What is the expression for Half-Life of a Second Order ReactionHere, I derive it from the integrated rate law.The answer is t 1/ (k A0)Ask me questions. 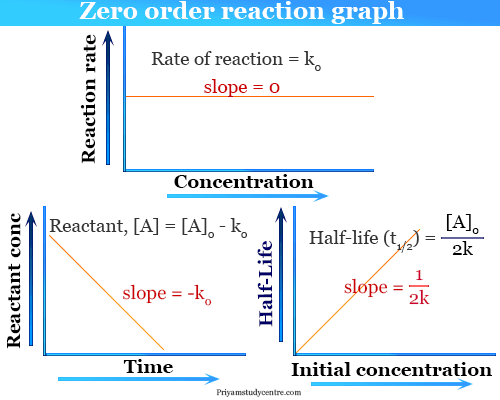
Notice how we cannot just pick # and call it good. (Clearly if #A = B#, then we are just doing (i).) Using #_0 ne _0#, we get a second order reaction of two first-order components #A# and #B#: Then, you can integrate each side with respect to the variable in question. The rate constant for a first order reaction whose half-life. What we can do is a separation of variables: It is not two #A# reactants reacting with a #B# reactant. Note that despite the fact you have two #A# reactants, you use the stoichiometric coefficient #\mathbf1#, not #2#, because #A# is reacting with itself. 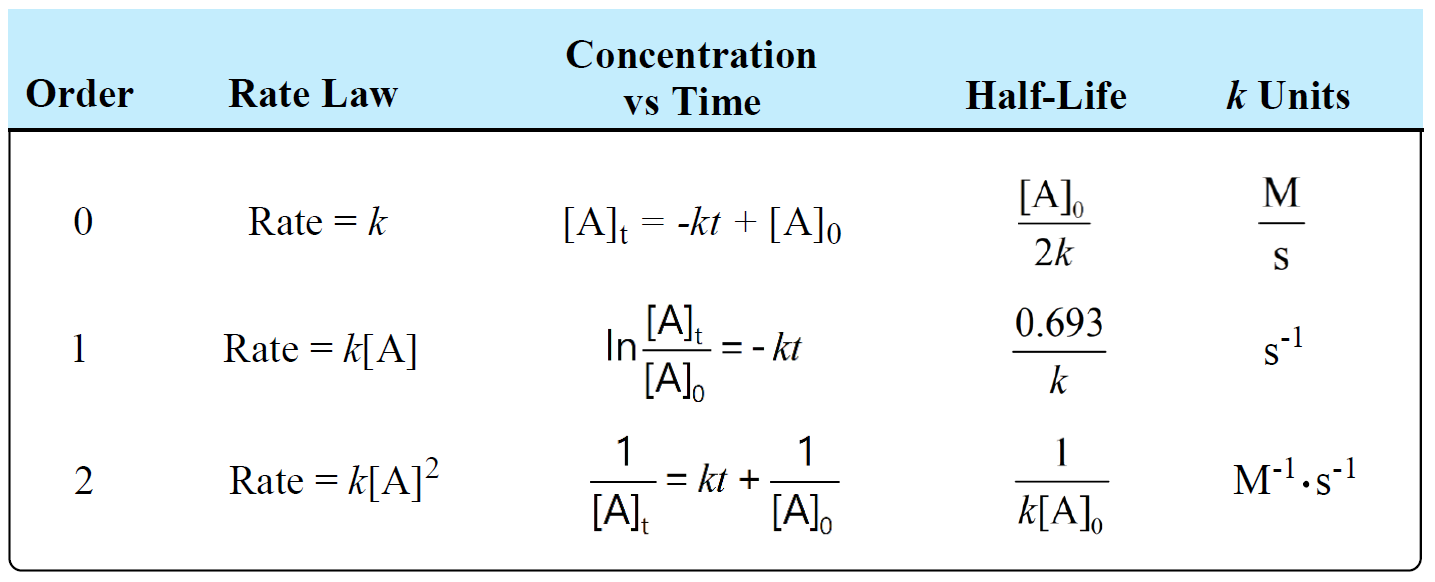
#r(t) = k = color(highlight)(k^2 = -(d)/(dt)) = (d)/(dt)# when order of reaction is first (n1) Half-life time for first order reaction is 300. Half life of second order reaction formula is defined as the time when the concentration of initial reactant in second order becomes half and is represented as T 1/2 1/ C A K second or Half Life of Second Order Reaction 1/ Reactant Concentration Rate Constant for Second Order Reaction. With equal amounts of two disappearing reactants, and calling the appearing product #C#, you have the rate law: For example, if m 1 and n 2, the reaction is first order in A and second order in B. Referring to the generic rate law above, the reaction is m order with respect to A and n order with respect to B. Solution Exercise 1.7.2 Second-Order Reactions Example 1.7.3: The Integrated Rate Law for a Second-Order Reaction Solution Exercise 1.7.3 Example 1.7.4: Determination of Reaction Order by Graphing Solution Exercise 1.7. e) There is not enough information to answer this question.Only through assuming that each reaction is an elementary reaction do we have The reaction orders in a rate law describe the mathematical dependence of the rate on reactant concentrations. In chemistry, the rate law or rate equation for a chemical reaction is a mathematical equation that links the rate of forward reaction with the concentrations or pressures of the reactants and constant parameters (normally rate coefficients and partial reaction orders). What is the activation energy? a) 0.014100 J b) 3.3602 × 1 0 + 4 J c) 0.0055 J d) 1.17 Jġ8) For problem 17, what is the rate constant for the reaction at 7 5 ∘ C ? a) 0.0055 sec − 1 b) 1.45 × 1 0 − 20 sec − 1 c) 0.033 sec − 1 d) 1.17 sec − 1 e) 3646.3 sec − 1ġ9) For problem 17, what are the half-lives at 2 5 ∘ C and 7 5 ∘ C, respectively? Assume that the initial concentration of reactants is 1 M. Calculate the time needed for the concentration of A to. The time needed for the concentration of A to decrease to one-fourth of its original concentration is: a) 150 sec b) 125 sec c) 75 sec d) 100 sec e) 50 secġ6) Given that the rate constant for a reaction is 1.5 × 1 0 10 at 2 5 ∘ C, what is its rate constant at 3 7 ∘ C ? Activation energy is 38 kJ / mol, and the value of R, the ideal gas constant, is 8.314 Jmole − 1 K − 1. The half-life for the second-order reaction of a substance A is 50.5 s when A00.84 molL1. 
14) The half-life for a second order reaction is 50 sec when 0 = 0.84 mol L − 1. The first-order reaction is given by, t1/2 0.693k which is independent of the initial concentration of reactant.Second order reaction, t1/2 1kA0. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
